02/04/2013
What Is Preimplantation Genetic Diagnosis?
ČSH
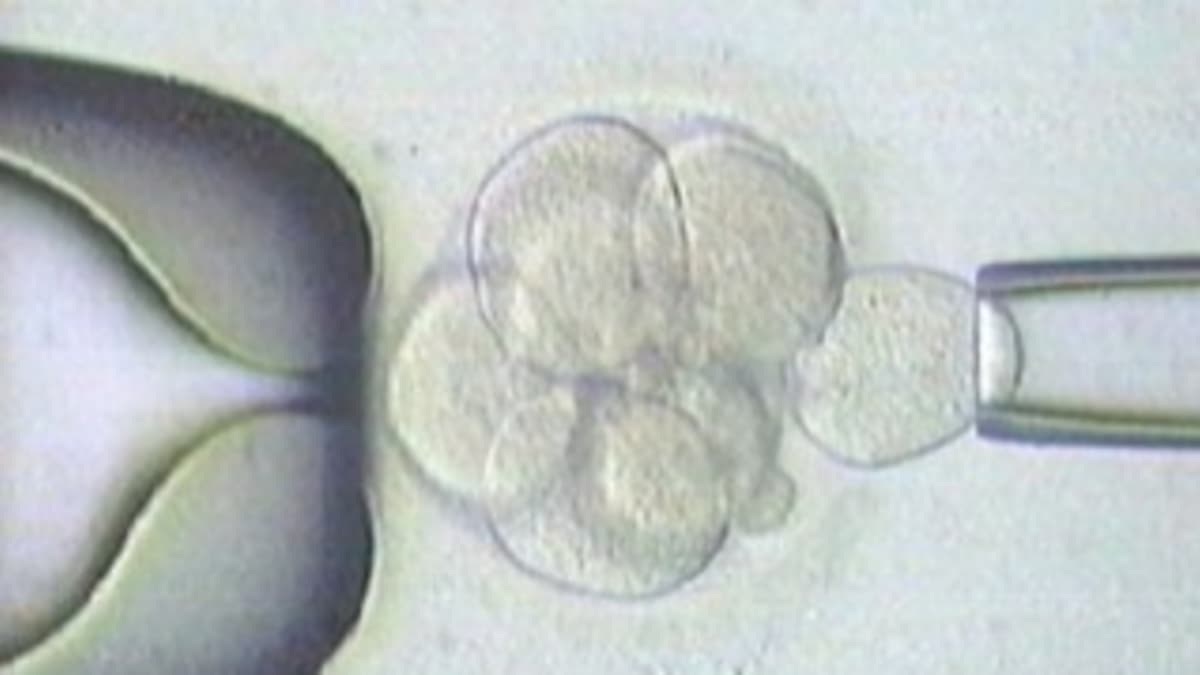
Carriers of rare hereditary diseases, including haemophilia, want to know when starting a family what the chances are of having a healthy child without the condition. They are offered two options: prenatal genetic diagnosis or preimplantation genetic diagnosis (PGD). PGD is a method that enables the selection of an embryo without a specific genetic burden. Its defining characteristic is that the embryo selection takes place before its transfer into the uterus.
The advantage is that the subsequent pregnancy is practically without risk that the child will carry the monitored genetic burden. There is no need to deal with the potential pitfalls associated with termination of pregnancy, which in practice mean both ethical dilemmas and health complications. Termination of pregnancy can also have an adverse effect on the couple's subsequent ability to conceive again. PGD is always combined with assisted reproduction methods and in vitro fertilization. The process begins with egg retrieval and their subsequent fertilization with the partner's sperm. One cell is then removed from the resulting embryos. This does not harm the early embryo in any way, and the removed cell is used for genetic testing. Only embryos in which the genetic test performed on the removed cell has ruled out the risk of the condition are then transferred into the mother's uterus.
Preimplantation genetic diagnosis is suitable for couples who already have one affected child, or who themselves suffer from a monogenic rare disease. Also for couples in whose families someone affected has occurred. The suitability of PGD for a specific couple is determined by a clinical geneticist.
PGD can currently be performed for any rare disease where we know which specific gene or mutations are responsible for the onset of the disease. This includes all forms of haemophilia where the precise genetic cause is known. For a carrier, each conceived boy faces a 50% risk of having haemophilia, and each conceived girl faces a 50% risk of also being a carrier. For such families, the PGD method is exceptionally suitable.
In the case of haemophilia, we also encounter cases where the X chromosome mutation arose in the egg during its development and the mother is not a carrier. In that case, it is not an inherited form of haemophilia, but a so-called de novo mutation. If the mother is not a carrier of the mutated gene, there is no concern that other siblings would develop haemophilia. However, a person with haemophilia caused by a de novo mutation typically passes it on to subsequent generations, meaning that all daughters born will become carriers.
The first important step is the preparation of PGD. A scheme must be prepared that reliably distinguishes the mutated gene; this involves DNA analysis of the couple and their closest family members. Subsequently, a precise and reliable procedure for examining the cell removed from the embryo can be designed. It can unambiguously distinguish the gene responsible for the onset of the disease from a gene without genetic burden, enabling the birth of a healthy child.
Simultaneously with the preparation and optimization of the genetic examination, preparation takes place for the couple to undergo assisted reproduction procedures, such as egg retrieval and in vitro fertilization.
During the PGD procedure itself, eggs are retrieved and fertilized, and after three days of embryo cultivation, one cell is removed. The DNA of the removed cell is used to determine what gene composition the embryo carries. Based on this, it is possible to say with near 100% certainty whether the tested embryo is either healthy or carries a genetic burden associated with future disease development. An embryo assessed as genetically suitable is subsequently transferred into the mother's uterus. If the couple has multiple embryos suitable for transfer, they can be very gently frozen and preserved for repeated transfer or for the future, should the couple wish to have another child.
The Brno-based Repromeda Sanatorium has extensive experience in this field, having used PGD since 2004. In addition to testing cells from early embryo stages (blastomeres), they also perform examinations of cells taken from later embryonic stages — blastocysts. In most cases, embryos are transferred into the mother's uterus at the blastocyst stage, with appropriate consideration for development quality. Thanks to continuous monitoring of embryo development, the most suitable embryos can be selected for transfer, thus achieving a high pregnancy success rate.
PGD for haemophilia and other rare diseases is fully covered by larger health insurance companies. The couple only co-pays for the preparation and optimization of the method for the first PGD cycle, which is always entirely individual for each couple and represents a co-payment of CZK 15,000. Additionally, the couple pays for procedures that are unavoidable during assisted reproduction but not covered by health insurance. These include, for example, intracytoplasmic sperm injection, extended embryo cultivation, and a co-payment for medications for the hormonal treatment needed for retrieving a larger number of eggs. The co-payment for these procedures currently amounts to approximately CZK 25,000.
Preimplantation genetic diagnosis cannot guarantee the birth of an absolutely healthy child, but it can definitively exclude the transmission of a specific genetic burden to the next generation. In such a case, the parental couple need not fear being forced to consider termination of pregnancy due to a known problem.
More information can be found at www.repromeda.cz
