11/03/2012
What Is Available — Updated September 20, 2016
ČSH

The range of therapeutic concentrates available in our country is relatively wide; we bring you an overview. All listed products are registered in the Czech Republic and can be used; the only exception currently applies to the product Wilate. Photographs of packaging and contents may change over time; they reflect the state as of the date of publication. The same applies to package sizes.
Advate (Baxalta) Recombinant human coagulation Factor VIII, third generation. It is manufactured using recombinant DNA technology in Chinese hamster ovary cells without the addition of any human or animal protein during cell cultivation, purification, or final formulation. It is stored in a refrigerator at 2 °C – 8 °C. During the shelf life, the vial with powder may be stored at room temperature (up to 25 °C) for a single period not exceeding 6 months. It is supplied in packages of 250, 500, 1000, 1500, 2000, or 3000 IU.

Beriate 250 IU/500 IU/1000 IU/2000 IU (CSL Behring) This medicinal product is manufactured from human plasma and contains 250/500/1000/2000 IU of human coagulation Factor VIII. Beriate is used for the treatment and prophylaxis of bleeding in patients with haemophilia A (congenital Factor VIII deficiency); it can also be used for the treatment of acquired Factor VIII deficiency. Shelf life is 3 years; store in a refrigerator, protect from freezing. In the Czech market, 1000 IU and 2000 IU packages are available.
One package with 1000 IU and with 2000 IU contains:
- 1 vial with powder
- 1 vial with 10 ml water for injection
One application kit contains:
- 1 transfer adapter with 20/20 filter
- 1 disposable 10 ml syringe
- 1 venipuncture set
- 2 alcohol swabs
- 1 non-sterile adhesive bandage

Factor VII (Baxalta) Coagulation Factor VII is manufactured from human plasma and is used for the treatment and prophylaxis of bleeding disorders caused by Factor VII deficiency or associated with Factor VII deficiency. It is stored at 2 °C to 8 °C in a 600 IU package.

Fanhdi (Grifols) Highly purified concentrate complex of coagulation Factor VIII and human von Willebrand factor, double-inactivated. It is indicated for the prophylaxis and treatment of bleeding in haemophilia A (congenital Factor VIII deficiency) and for the prophylaxis and treatment of bleeding in von Willebrand disease (vWD) when treatment with desmopressin (DDAVP) alone is ineffective or contraindicated. The product can also be used in the treatment of acquired Factor VIII deficiency. First registered in 1997, it can be stored at room temperature up to 30 °C for up to three years. It is supplied in packages containing Factor VIII coagulationis 250, 500, 1000, or 1500 IU and Factor von Willebrand humanus 300, 600, 1200, or 1800 IU.

Feiba (Baxalta) A product manufactured from human plasma that supports blood clotting even when individual coagulation factors are reduced or absent. It also contains coagulation Factors II, IX, and X, as well as activated coagulation Factor VII. It is used for the treatment and prophylaxis of bleeding in haemophilia A and B with inhibitors. It can also be used for the treatment and prophylaxis of bleeding in non-haemophilic patients who have developed inhibitors against Factors VIII, IX, and XI. It is stored in a refrigerator at 2 °C – 8 °C. Within the stated shelf life, the product may be stored at room temperature (max. +25 °C) for a period of 6 months. It is supplied in 500 and 1000 IU packages.

Haemate P 500 IU, 1000 IU (CSL Behring) Haemate P is manufactured from human plasma and contains human von Willebrand factor and human coagulation Factor VIII. Haemate P is used for the treatment or prevention of bleeding from von Willebrand factor deficiency during surgical procedures when treatment with desmopressin (DDAVP) alone is ineffective or contraindicated. Haemate P is used for the prevention or control of bleeding in Factor VIII deficiency in the blood. This medicinal product may also be used for the treatment of acquired Factor VIII deficiency and for the treatment of patients with antibodies against Factor VIII. Haemate P is supplied in 500 IU and 1000 IU packages. Store at temperatures up to 25 °C, protect from freezing.
500 IU package:
- 1 vial (hermetically sealed under vacuum) with dry substance
- 1 vial with 10 ml water for injection
- 1 transfer filter set 20/20
- 2 disinfection alcohol swabs
1000 IU package:
- 1 vial (hermetically sealed under vacuum) with dry substance
- 1 vial with 15 ml water for injection
- 1 transfer filter set 20/20
- 2 disinfection alcohol swabs

Helixate NexGen 250 IU, 500 IU, 1000 IU (CSL Behring) Helixate NexGen contains the active substance recombinant human coagulation Factor VIII (octocog alfa). Helixate NexGen is used for the treatment and prophylaxis of bleeding in adults, adolescents, and children of all age groups with haemophilia A (congenital Factor VIII deficiency). This product does not contain von Willebrand factor and therefore should not be used in von Willebrand disease. Store in a refrigerator (2 °C – 8 °C), protect from freezing. Each Helixate NexGen package comes with medical devices for reconstitution and administration (application kit).
Additional package contents:
- 1 transfer filter adapter 20/20 [Mix2Vial]
- 1 venipuncture set
- 1 disposable syringe (5 ml)
- 2 disposable alcohol swabs

Immunate (Baxalta) A complex of coagulation Factor VIII and von Willebrand factor manufactured from human plasma. It is used for the treatment and prevention of bleeding in haemophilia A or acquired Factor VIII deficiency and von Willebrand disease with Factor VIII deficiency. Store at 2 °C – 8 °C; during the shelf life, it may be stored at room temperature up to 25 °C for a maximum single period not exceeding 6 months. It is supplied in 250, 500, and 1000 IU packages.

Immunine (Baxalta) Coagulation Factor IX concentrate, used for the treatment and prevention of bleeding in congenital haemophilia B. Store in a refrigerator at 2 °C to 8 °C. During the shelf life, it may be stored at temperatures up to 25 °C for a maximum of 3 months. It is supplied in 600 and 1200 IU packages.

Kogenate (Bayer) Second-generation recombinant coagulation Factor VIII. It is produced using bacteria through so-called recombination, whereby a human gene carrying information about how Factor VIII should be structured is inserted into a specific bacterium, which then produces it. Kogenate Bayer is administered as an injection into a vein. The volume of solution that needs to be injected during a single application is only 2.5 ml; the powder containing Factor VIII is mixed with solvent warmed to a temperature up to 37 °C. Everyone has the opportunity to practise correct administration using the Baycuff training pouch. Kogenate Bayer is manufactured in Berkeley, California, and was introduced to the market in 2000.

Mononine 500 IU (CSL Behring) Mononine contains human coagulation Factor IX. It is used for the prevention or control of bleeding caused by congenital Factor IX deficiency (haemophilia B) in the blood. Store in a refrigerator (2 °C – 8 °C), protect from freezing. The box with 500 IU contains: 1 vial with powder, 1 vial with 5 ml water for injection. Each Mononine package comes with an application kit containing:
1 transfer adapter with 20/20 filter 1 disposable 10 ml syringe 1 venipuncture set 2 alcohol swabs 1 non-sterile adhesive bandage

NovoSeven (Novo Nordisk) Recombinant activated coagulation Factor VII. It is used as so-called "bypass" therapy, which enables effective blood clotting even without the presence of certain natural coagulation factors, bypassing the points in the coagulation process where coagulation factors are missing. Administered as an injection into a vein, it is used for the treatment of haemophilia A and B with inhibitors against coagulation Factors VIII and IX, where the inhibitor arose as an immune reaction of the body to administered substitution products, and for Glanzmann thrombasthenia — a rare platelet function disorder. In addition, NovoSeven is also indicated as substitution therapy for congenital coagulation Factor VII deficiency. It is stable at temperatures from 2 to 30 °C for up to 3 years, in ampoules of 1 mg, 2 mg, and 5 mg.

Octanate (Octapharma) Highly purified concentrate of human coagulation Factor VIII stabilized with von Willebrand factor, treated with two viral inactivation methods. Factor VIII is bound by natural stabilizer vWF in a ratio of vWF/FVIII of approximately 0.4, therefore no additional stabilizers are needed. Octanate is used for the treatment of bleeding in all types of haemophilia A and for eradication of Factor VIII inhibitors. It is supplied in packages containing 250, 500, and 1000 IU. Octanate was first registered in Germany in August 1998 and subsequently in 67 other countries.

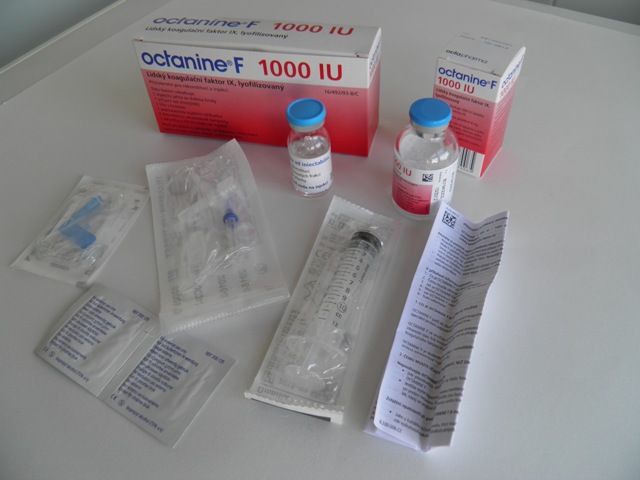
Octanine F (Octapharma) Highly purified concentrate of human coagulation Factor IX, treated with two viral inactivation methods. It is intended for the treatment and prophylaxis of bleeding in haemophilia B. No denaturing processes occur during production; the physiological functionality of the Factor IX molecule is preserved. Octanine F does not contain mouse or hamster protein impurities, and therefore its administration is not contraindicated in cases of allergy to these. The product may be stored throughout its shelf life at temperatures up to 25 °C, in packages of 250, 500, and 1000 IU.
Recombinate (Baxalta) First-generation recombinant concentrate used for the treatment or prevention of bleeding in haemophilia A. It does not contain von Willebrand factor and is therefore not used for the treatment of von Willebrand disease. It contains 1.5 mmol sodium per dose, which should be taken into account when treating patients on a sodium-controlled diet. During the shelf life, it may be stored at 15 – 25 °C for up to six months. It is supplied in three strengths: 250, 500, or 1000 IU.

Willfact (LFB/Sobi) The only nearly pure concentrate of human von Willebrand factor (1,000 IU per vial). A unique 3-step purification manufacturing process (including nanofiltration) ensures complete viral inactivation. Willfact is intended primarily for the prophylactic treatment of patients with von Willebrand disease. The product must be stored in its original packaging to protect it from light, at temperatures up to 25 °C. In the Czech Republic, Willfact has been registered since 2011 and is available on the basis of approval for exceptional reimbursement by the relevant health insurance company. It is currently supplied to the Czech Republic in German packaging with a Czech label and Czech patient information leaflet.

Wilate (Octapharma) It is registered in the Czech Republic but does not yet have an established reimbursement, so it is not yet available. It is the first so-called highly purified concentrate of von Willebrand factor and coagulation Factor VIII (vWF/FVIII) with two viral inactivation steps incorporated into the manufacturing process. The result is achieving a 1:1 ratio of von Willebrand cofactor to FVIII activity, a ratio similar to that in normal human plasma. The product does not contain albumin. It is manufactured exclusively from large pools of human plasma obtained at plasma donor centres approved by the U.S. Food and Drug Administration (FDA), which, due to its high viral safety profile, has classified it as an "orphan drug." The product is registered in more than 40 countries worldwide, including most EU countries, as well as in the USA, Canada, Australia, Brazil, and Russia.
